Stratum Corneum Diffusion Testing shows how quickly and how much of an active ingredient can cross the top layer of the skin. This data supports formulation work on creams, gels, ointments, and patches, and is treated as an important signal of product performance. In simple terms, it should tell us how fast and how much of an ingredient can cross the skin barrier. Yet many teams find that their diffusion data is unstable or hard to reproduce. Why do results look so different from run to run - and what is quietly limiting accuracy behind the scenes?

Why Accuracy in Stratum Corneum Diffusion Testing Is So Hard to Achieve
On paper, Stratum Corneum Diffusion Testing should be simple: place your membrane, load the formulation, collect samples over time, and read the curve. In reality, two runs carried out under "the same" conditions can tell very different stories. Data from different analysts, different days, or different labs often refuses to line up. Projects slow down, and teams waste time debating whether the formulation is unstable or the method is unreliable.
A big part of this problem comes from how much is still done by hand. Many setups rely on technicians to assemble each Franz cell, adjust the temperature bath, remove bubbles, and pipette samples at fixed time points. Every action introduces a chance for small deviations. Sampling a minute late here, a minor temperature shift there, or a thin bubble trapped beneath the skin can all bend the release curve without anyone noticing.
The membrane itself adds another layer of uncertainty. Human, animal, and artificial skin models do not behave in exactly the same way. Changes in skin thickness, hydration, or preparation can tilt the apparent permeation rate. When this natural variability meets an unstable cell - maybe a slightly leaking joint or poor stirring - the combined effect makes Stratum Corneum Diffusion Testing results feel more like "suggestions" than hard data.
For teams developing creams, gels, ointments, and patches, this is more than a technical nuisance. It means repeating runs, discarding suspicious data, and holding long internal discussions about what to trust. In the end, the lack of confidence in diffusion data becomes a real obstacle to decision-making and slows the path to market.
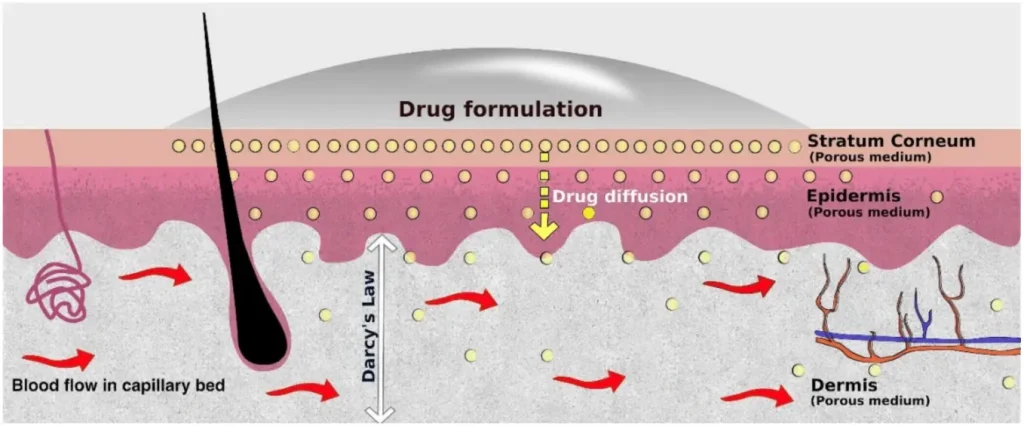
The Hidden Error Traps Behind Unstable Diffusion Data
Many issues in Stratum Corneum Diffusion Testing are subtle. They do not come from the active ingredient or the formulation design, but from the way the test system behaves minute by minute.
Operational Details That Quietly Skew Results
Even when analysts follow SOPs carefully, small operational factors can accumulate and distort results:
✅Micro air bubbles stay in the diffusion cell and partially cover the membrane surface
✅Sampling intervals drift, changing the shape and slope of the release curve
✅Temperature varies slightly from cell to cell, affecting permeation rates
✅Tiny leaks in the glassware or joints slowly dilute the sample in the receptor medium
✅Cross contamination appears when several cells share tubing or pumping elements
Individually, each factor seems minor. Together, they can severely limit the accuracy of Stratum Corneum Diffusion Testing. When comparing two formulations, the one that "performs better" may simply have been measured in a slightly more stable cell. This is a frustrating situation for any R&D team that needs clear, data-based answers.
Why Smarter System Design and Automation Matter?
To move past these limits, the hardware and automation behind the method need to do more of the heavy lifting. This is where the Raytor RT814 Automated Transdermal Diffusion System steps in as a purpose-built solution.
RT814 is a fully automated, 14-position Franz cell system built around the real pain points of diffusion testing. Instead of asking operators to manually tilt cells and visually check for bubbles, its mechanical design automatically tilts the diffusion cell and removes trapped air in real time. The membrane stays properly exposed, and one common source of error is continuously managed by the system, not by the operator's eyes.
The diffusion cells are made as an integrated glass structure to reduce the chance of leaks during the run. Each cell has its own dedicated pipeline and pump, which sharply lowers the risk of cross contamination. Independent temperature control for every cell helps keep conditions consistent even when different formulations or methods are tested in parallel.
For labs working under European Pharmacopoeia 2.9.4 and USP <1724> expectations, this kind of design directly supports more robust Stratum Corneum Diffusion Testing. It becomes easier to argue that differences in profiles arise from the formulations, not from a temperamental test setup.

How the RT814 System Builds Trustworthy Diffusion Profiles
In practice, accuracy is less about drawing a "nice" smooth curve and more about believing that the curve reflects how the product truly behaves. The RT814 system is designed to bring Stratum Corneum Diffusion Testing closer to that ideal in day-to-day work, not just in theory.
The platform combines an integrated diffusion cell block, an automatic sampling module, and a medium thermostat into one coordinated system. Automation reduces the number of manual touchpoints, which in turn reduces the opportunities for small but impactful differences between tests. Two independent groups of diffusion cells can be run at the same time, so teams can compare two formulations, two methods, or two conditions in a single study without interference between channels.
Sampling flexibility is another practical advantage. RT814 supports both partial and full sampling, allowing method developers to match their protocol to in vitro release or in vitro permeation studies in line with USP <1724>. The system can log sampling operations and conditions in real time, creating a clearer audit trail and simplifying method transfer or regulatory review.
From a cost perspective, one of the most painful issues in Stratum Corneum Diffusion Testing is rework. Unstable or questionable runs force repeats. Each repeat consumes reference materials, analysts' time, and instrument capacity. By controlling bubbles, leaks, contamination, and temperature more tightly, RT814 helps reduce the number of runs that end up being labeled as "outliers" or "not usable".
Looking at the bigger picture, stronger diffusion data also improves communication with regulators and external partners. When results come from a system that aligns with EP and USP requirements and is supported by thoughtful automation and engineering, you are not just presenting numbers. You are presenting a method that is easier to defend and easier to reproduce across sites.
Call to Action
If your team is still wrestling with inconsistent Stratum Corneum Diffusion Testing curves and too many repeat studies, it may be time to upgrade more than just the formulation. Explore how the Raytor RT814 Automated Transdermal Diffusion System can help you generate cleaner, more reliable diffusion data for creams, ointments, patches, and gels - and transform diffusion testing from a clinical bottleneck into a confident, decision-driving tool.